|
A σ-bond is symmetrical in terms of rotation about the bond axis in this formal approach. Sigma bonding is best defined for diatomic molecules using the language as well as strategies of symmetry groups. In chemistry, sigma bonds ( σ bonds) are perhaps the most basic type of covalent chemical bond. Polymer chain backbones, as well as thermosetting polymer cross-links, contain it as well. Covalent bonding is the common type used in silicate ceramics and glasses.Covalent bonding can be particularly stiff, resulting in such a material with the properties listed below Electrons move among atoms in a three-dimensional structure of remarkably directed bonds. When atomic orbitals overlap, covalent bonds will be formed. 
At least one electron from each atom will be shared by two covalently bonded atoms. In covalent bonding, sharing electrons between the surrounding atoms results in a stable electronic structure. Based on the type of overlapping, covalent bonds are categorized as sigma bonds or pi bonds. The shared number of bonds varies with the number of electrons. Both are widely used in molecular orbital theory to predict the outcome of molecules. Both sigma and pi are derived from Greek letters. 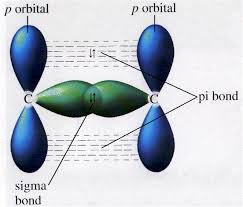
Sigma bonds are much stronger than pi bonds. The atomic orbitals of Pi and Sigma Bonds will undergo overlapping, which is the main difference between them. The two most common chemical bonds are sigma bonds and pi bonds. Chemical bonds can be of various shapes and sizes. Next → ← prev Difference Between Sigma and Pi BondĪ chemical bond is a strong force that connects two atoms.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
